Summary
- How can release be accelerated via CAPA?
- Release driven and controlled by visual management
- Electronic batch record: the opportunities… and the pitfalls to be avoided
- The challenge of digitalizing production records
- AQU@Sense MB: A technological evaluation for « on-line » pharmaceutical water analysis
- Individual closed isolators for cell therapy
- Evaluation of a Quaternary Ammonium Ready To Use (RTU) Disinfectant and Hydrogen Peroxide/Peracetic Ready To Use (RTU) Combination Sanitization Regimen for Cleanroom Start-Up
The challenge of digitalizing production records
We are in the era of the fourth industrial revolution where new technologies play a central role in the creation of intelligent factories that adapt more easily to production necessities and processes. In this context, increasing importance is given to Artificial Intelligence (AI), to robotic process automation (RPA) and to processing metadata…

Although projects with this type of complex technology are emerging in the pharmaceutical industries, the records that are the day-to-day medium of operational traceability and batch release are still very often in paper format. This technological paradox raises the question: what are the difficulties in digitalizing these records? What are the benefits to be drawn from this?
The purpose of this article is to inform you about a transition of records (batch record, preparation sheets, cleaning documents) from a paper version to a digital version on a biotechnological production site of average size (commercial product, around 100 persons).
1- Context
Two main types of document are used to comply with Good Manufacturing Practices and to produce in a secured environment: instructions and records /reports. These documents are generally managed with EDM software (Electronic Document Management). However, although the master record documents are also in an EDM, they are still often completed manually.
Managing document completion manually creates different problems:
• Risk of completion oversights and errors particularly in calculations;
• Transport of documents from printing areas to production areas, reviews and archiving and risk of loss / deterioration of documents;
• Transfer of manual data into computerized systems in order to analyse data;
• Difficulty in accessing documents for consultation / search for data / investigations;
• Onerous nature of the archive management process.
These problems translate negatively into different indicators such as long release time frames or low “right first time” indicators for documentation.
It is in this context that a digitalization project was set up with the creation of a multidisciplinary working team involving production, quality, qualification and the IT department.
2. Definition and choice of the digital solution
The first constraint in setting up this project was financial: the site did not have sufficient resources to install a general production management software application such as MES. The overall budget allocated for setting up the project was €100m, which determined the definition of need.
The search for this digital solution was facilitated by the presentation of a catalogue software application by one of our suppliers that had already developed the site equipment supervision system.
This CiiDOC software (https://www.actemium-process-automation.com/) is based on Word documents (see Figure 1), in which tags with controlled fields of different types (date, signature, time, itemization, checkbox, calculation, attachment etc) are inserted.
After an approval cycle, this document is available for the user who creates a production order with a unique identifier then completes the tags digitally. The information completed is recorded in real time in a database (SQL Server) whose use is customizable. Once the production order is completed, a PDF version can be generated to facilitate document circulation and access for persons who do not have access to the software.
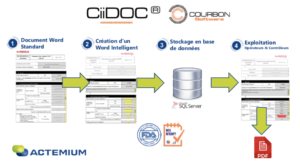
The software was designed in such a way so to comply with the regulations on the use of digital documents and electronic signatures (FDA 21 CFR Part 11), which represented an essential prerequisite for GMP validation of the system. In addition, CiiDOC is compatible with a user management system via Windows Server Active Directory (AD) which is commonly used and in place on the site. User rights management is based on the setting up of Windows groups in the AD and is therefore completely customisable.
A test environment was then put in place to check that the software could respond correctly to the specific features of our documents and document management process while complying with the quality procedures in place. This test phase was essential to understand how the software functioned, to evaluate the workload involved in installing tags in documents, to define the elements to be configured such as access groups and also to run demonstrations to facilitate acceptance of the change by the different project stakeholders.
In parallel with these tests on the software, a document overhaul was carried out on batch records, solutions/buffer preparation sheets and cleaning documents (see académie efficace website: https://www.academie-de-la-qualite-efficace.fr/). These documents were standardized with a user-friendly column structure and simplified in order to retain only relevant data, which represented the elimination of 48 % of initial batch record data. They were used in paper format while the digital solution was being developed and convinced all users of their efficacy. This ergonomic structure then facilitated the configuration of documents in CiiDOC, as each box defined in the column structure contained a tag. There was therefore no structural change to documents during the transition to CiiDOC, just a configuration of tags according to the nature of the data item to be collected (definition of tag type, any limitations, display etc).
3. Difficulties and challenges of implementation
Once the needs were defined and the solution chosen, more than one year went by before the first digitalized records were used in production. Project implementation was long and complex despite the definition of clear objectives and the setting up of a multidisciplinary working group.
The first difficulty encountered was a problem of communication and project management with users, the IT department and the supplier, which caused a delay in implementing actions. The major cause of this problem was the language difference between the computer scientists and users, which created fruitless, frustrating exchanges for all parties, including the supplier. The second cause related to the organization of large groups, which generally have a general, decentralized IT department. This IT department management system demands an anticipation of actions sometimes incompatible with the needs inherent in the setting up of projects of this kind and also creates misunderstandings with regard to the specific features of local IT. In addition, because of the desire to simplify the support provided long-term and the creation of standard catalogues, the possibilities of putting in place solutions outside the defined framework are limited. It is therefore essential to have a person who acts as intermediary between the IT department, the supplier and local businesses, who is capable of translating the concrete needs of the project into technical specifications and conversely, of asking the right questions that will allow streamlined project management.
The second major difficulty lay in managing the software validation phase between users, the Qualification team and Quality Assurance. To illustrate this point, let us take the example of the drafting of specifications by the user (URS – User Requirement System), the gateway to the validation cycle, which lasted for several months. This step is crucial as it determines what happens after qualification: a poorly identified need has an impact on the tests carried out subsequently. The document had to be redesigned several times because of a lack of understanding and knowledge of the level of detail expected (requirements concerning data integrity for example). One of the causes of the difficulties encountered during this validation can be explained by the “silo” working method of these departments which are located physically in different buildings and operate independently with little exchange of information apart from project meetings. It therefore appears necessary to provide for more time to promote department interfacing. The ideal would be to plan blocked-out working sessions sufficiently in advance to move forward simultaneously and to promote synergistic working.
Finally the last difficulty was to find the support necessary among future users in production teams which had other priorities and therefore little time to devote to this subject. Document configuration was thus entrusted to a consultant and the digitalization project stayed in a “black box” for many people before its final implementation. The risk of operating in this way is of not pinning down all user needs and of validating a configuration that is not completely appropriate for their use. The change management process and continuous validation cycle process then often holds up subsequent changes and leads to frustration which could have been avoided with more involvement from the beginning of the project.
In taking a more global view of difficulties relating to this digitalization project, the real question to be asked is perhaps: was the project management method used really adapted to digitalization?
Projects in the pharmaceutical world are classically managed “in cascade”: all needs are defined from the beginning and the steps are then performed one after the other until final implementation.
Because of this fact, the “subject” is a fixed element while resources and time are adjusted until the fixed objective is achieved. This mode of operation is quite suitable for most projects and even for a change of software version, for example, as the subject is clear, the risks easily identifiable and a detailed schedule of actions can be constructed easily. However this type of management does not leave flexibility for a project involving a significant amount of configuration, such as the setting up of digitalized records, and raises a problem if a need has been poorly defined or is new.
There are other project management methods, such as the Agile/Scrum model which operates in iterative, incremental cycles called “sprints”. The advantage is then to work in a team with fixed time frames and to gradually construct a concrete solution that will best suit user needs. This way of operating is compatible with the validation life cycle which is just accelerated but demands a major change in skills, resource allocation by project leaders and time management. The real challenge is perhaps therefore to set up multidisciplinary teams dedicated to Agile/Scrum methodology to enter the digital revolution with ease?
4. The benefits of installation
Despite the difficulties encountered during project implementation, user feedback is positive.
Below are extracts from the feedback of two technicians who work in the media and buffers preparation department (see Figure 2):
“For the 7 years that I have been here, people have always commented on my handwriting, thanks to CiiDOC, this problem no longer exists!”
“The system allows calculations to be carried out automatically therefore this prevents us from making mistakes in calculating times or expiration dates (especially when we have to add 47 days)”
“Through digitalization, we avoid problems of looking for sheets in different buildings and we can access information more easily when we need to consult it”.

Users are all the same disappointed that digitalization is not complete with a direct link between the software and the different measurement devices and equipment, which could be a future development in collaboration with an automation team.
In addition to the obvious advantages such as the reduction in review time resulting from the presence of automatic checks and centralization of data which provides optimal document traceability, digitalization also opens up areas for improvement.
Through retrieval of the content of all tags in real time in a database, it is then possible to program targeted retrievals for all types of application: quality and statistical analyses for continued process verification (CPV) or indeed performance with the creation of indicator tracking dashboards.
5. Conclusion
Paper-based management of production records makes their completion and review complex, which impacts release deadlines and documentation “right first time” indicators. The introduction of digitalization of these records in classic project mode is complex because of numerous difficulties particularly relating to the communication and management of IT issues, validation, and production support. However another more suitable project management method such as “Agile/Scrum” would facilitate working in a team and would provide more flexibility in order to respond to user needs in the best way possible.
This digitalization improves the traceability of records and simplifies the completion and review of documents but also opens other prospects for improving the management of quality or performance processes. A challenge which should be taken up!
Partager l’article

Aline Graciet – Merck
Aline Graciet est ingénieur en biotechnologie diplômée de l’ENSTBB (Bordeaux). Elle a commencé son parcours professionnel en tant que consultante Assystem en Belgique où elle a travaillé pour Baxter et GSK. Elle a été rapidement attirée par la philosophie LEAN dès le début de son parcours par envie d’initier et d’accompagner des changements qui faciliteraient les processus pharmaceutiques souvent inutilement complexes et historiquement figés. Responsable Excellence Opérationnelle sur le site Merck Healthcare de Martillac (Gironde, France) depuis 2019, elle développe ses compétences digitales afin de moderniser et simplifier la gestion de la documentation.
aline.graciet@merckgroup.com


