Sommaire
- When the pharmaceutical chain invents its own standards for its invisible links?
- Carriers & Good Distribution Practices for Medicines (BPDG). When every kilometer matters for quality.
- The importance of cleaning and disinfection procedures in controlling contamination in cleanrooms.
- The Impact of Next Generation Sequencing (NGS) on Quality Control of Pharmaceutical Products
- From Contamination to Continuous Improvement: A Method for Investigating Microbiological Deviations in the Pharmaceutical Industry
- Economic justification for automation & robotics solutions in pharmaceutical microbiology quality control
- Design and Operation of a Microbiology Laboratory: The Collaborative Approach of the GIC A3P Microbiology Laboratory.
- Best practices for using a Class II biological safety cabinet in a microbiology laboratory.
- Waste mapping
Design and Operation of a Microbiology Laboratory: The Collaborative Approach of the GIC A3P Microbiology Laboratory.
The design of a microbiology laboratory goes beyond simply arranging laboratory benches; it serves as a critical barrier against cross-contamination and ensures the reliability of analytical results.
Developed through the collaborative efforts of the GIC A3P Microbiology working group, this GST titled “Best Organizational Practices for a Microbiology Laboratory” offers a structured approach to designing an optimal microbiology laboratory layout.
From workflow management to the identification of critical areas and workplace ergonomics, this article outlines the fundamental principles for designing and/or optimizing your microbiological quality control facilities, as detailed in GST No. 23 (available online in the A3P member portal)
1. Fundamental Design Principles
The physical layout of the laboratory must meet strict requirements to ensure the safety of personnel, the environment, and the products.
The guide recommends ample work surfaces to prevent clutter, which can lead to contamination.
- It is recommended to allocate 15 to 20 square meters of floor space per technician, including equipment and workbenches.
- Ideally, each operator should have 6 meters of bench space, including equipment.
- The distance between technicians is also standardized to ensure smooth workflow: 1.5 meters when working back-to-back and 1.2 meters when working side-by-side.
The choice of finishes is crucial. Surfaces (walls, floors, furniture) must be:
- Smooth and non-porous for easy cleaning.
- Resistant to detergents and disinfectants.
- Non-slip for employee safety.
Light fixtures should be recessed to prevent dust buildup.
With regard to environmental conditions, the laboratory must maintain a temperature between 18 and 25°C.
2. Clothing and Hygiene
The laboratory must be a restricted-access area.
The design is tailored to the specific functions of each area:
- Standard Microbiology Area: Dedicated lab coat with long sleeves that fit snugly at the wrists, and dedicated lab shoes.
- Log Handling Area: Additional protective clothing, safety goggles, and gloves.
- Class A/B Areas: Full-body suit, mask, boots, goggles, and gloves. The entire outfit must be sterile.
- Specific Areas (such as PCR labs and sterility testing isolators): Appropriate protective clothing must be worn (hairnet, gloves, mask).
Hand hygiene is considered the first line of defense against infection, through handwashing and/or the use of hand sanitizer.
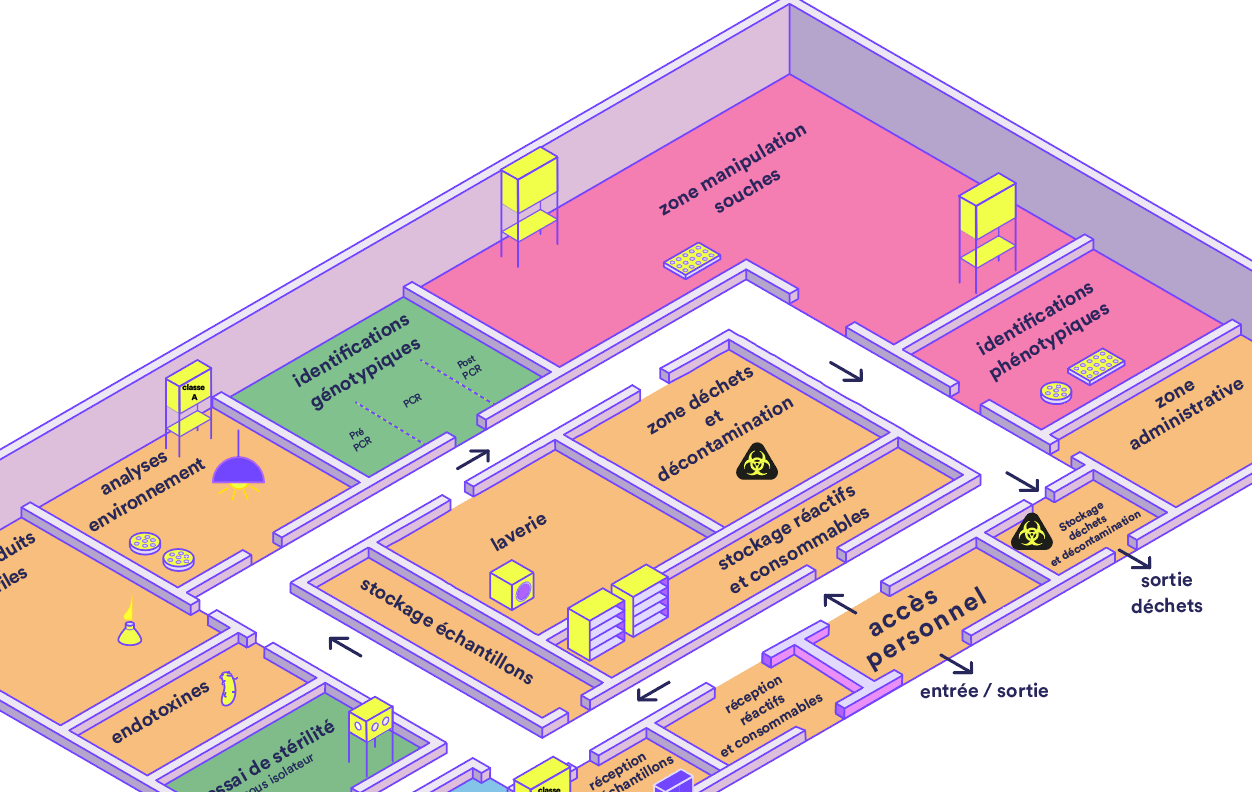
3. Workflow Management
The standard layout of a microbiology laboratory is based on the principle of one-way traffic and a strict separation of personnel, equipment, consumables, media, and materials.
A laboratory must be divided into separate zones based on the activity:
1. Receiving and Storage: Areas designated for receiving samples and supplies, located at the laboratory entrance.
2. Routine Testing: Separate rooms designated for specific activities and types of products being tested.
3. Critical areas:
- PCR: The pre-PCR and post-PCR areas must be kept separate to prevent contamination.
- Sterility testing: Must be performed in a Class A isolator or under Class A laminar flow in a Class B area (with a Class C/D airlock).
4. Strain Management: A dedicated area for handling strains and conducting related tests.
5. Auxiliary areas: Laundry room, autoclave, culture medium storage, and waste disposal area.
6. Administrative area: This area must be separated from the laboratory work areas (no laboratory work may be performed here, and no culture media may be stored here).
Waste management follows a specific process: outgoing waste must be kept separate from incoming materials to minimize the risk of contamination, using clearly labeled medical waste containers.
4. Protective Equipment: PSM vs. Laminar Flow
The selection and placement of microbiological safety cabinets are important factors.
- Type II Biological Safety Cabinet (BSC): It simultaneously protects the operator, the product, and the environment, particularly when handling Class 2 agents.
- Laminar flow hood: It is designed solely to protect the product. Therefore, it should not be used when handling hazardous materials, as it does not protect personnel.
To ensure their effectiveness, this equipment should not be placed near drafts or high-traffic areas (keep a distance of at least 1 meter).
Conclusion
Optimizing the layout of a microbiology laboratory involves striking a balance between regulatory compliance (ISO 7218, European Pharmacopoeia), operational efficiency, and available floor space.
By following the recommendations of the GIC A3P, manufacturers can ensure the safety of their testing processes while providing a safe and ergonomic work environment, thereby helping to streamline workflows and improve overall operational efficiency.
However, conducting a 5M risk analysis remains essential for adapting these general principles to each specific site.
To learn more about these concepts, we invite you to consult the full text of Scientific and Technical Guide No. 23 (GST No. 23 _2025), titled “Microbiology Laboratory: Best Practices & Organization,“ now available online in the members’ area. This guide serves as a resource for both the design of new facilities and the optimization of existing laboratories.
It includes key operational documents, such as:
- Strict health and safety guidelines.
- Detailed guidelines for handwashing, a cornerstone of cross-contamination prevention.
- A standard laboratory layout showing the division into zones and the recommended cladding for each activity.
Download the guide!
Finally, to continue the discussion on these topics and share feedback with the GIC experts, we look forward to seeing you at the A3P Microbiology Conference, which will be held in Strasbourg on June 24 and 25, 2026.
Share
Marina DAUBARD
Amélie FREJ
Séverine GAUBERT






