Summary
- Analytical Quality by Design: the required integration for Quality by Design
- Design of a production isolator. From user need to realization.
- Advanced vaporized H2O2 decontamination technology for pharmaceutical isolators. Reduction of H2O2 decontamination cycle time using direct injection nozzles.
- Secure the containment of your gloves
- Isolator Technology and Automation Enhanced Contamination Control in the Manufacture of Cell and Tissue Culture Derived Regenerative Medicine Products.
- The European approach to disinfectant qualification.
It is an absolute truth that all sterile production line installations must be associated with an appropriate means of protection of the product and the environment. When this choice relates to isolator technology, very particular attention must be paid to its design. This must be based on a perfect match between user need and the current state-of-the-art of this cutting-edge technology, culminating in an optimised solution for future production.
The installation of a new injectables production line at the Aspen site at Notre-Dame-de-Bondeville (76) is an example. The site, located in Normandy close to Rouen, has specialised in the production of prefilled syringes for many years and produces almost 118 million syringes each year destined for more than 30 international markets including the USA.
Key elements in the decision to achieve an optimal solution for future production.
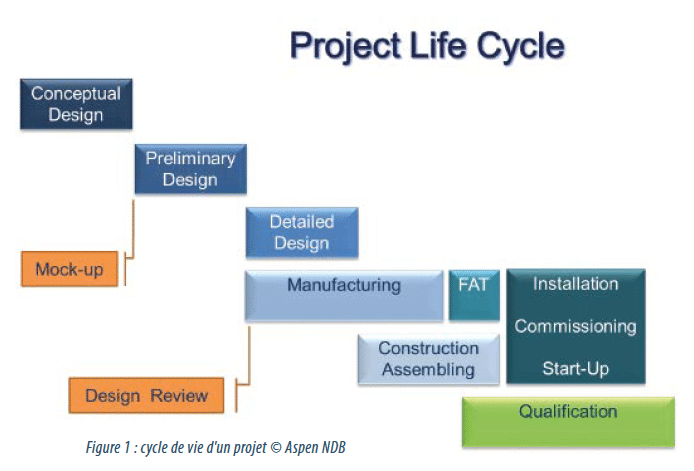
Of course, the commissioning of a new line is the result of a project process governed by a succession of steps and specific tasks, which must be strictly observed.
From the initial idea to installation and commissioning, the different steps of the project are used to refine an increasingly precise requirement. At the end of the process, the various contributions will enable the procurement of equipment which as far as possible represents an optimised solution.
Constructing the specifications
What exactly do we want? The existence of a previous, relatively recent production line on the Aspen site, with a use similar to that of the future line, facilitates the construction of specifications. The collection of information regarding past events, incidents that have arisen, improvements made, as well as regulatory changes, will inform these specifications at a very detailed level.
In the specific case of the design of an isolator, several fundamental aspects must be taken into account when writing the specifications.
In addition to the internal characteristics and regulatory standards, product and/or process characteristics must also be taken into account, as well as the equipment that will be connected to the isolator. It is in these areas that effort must be concentrated. Of course, the ergonomic and safety constraints must not be forgotten.
Collecting information on the specific needs of users
In order to refine our specifications for this project, staff who worked on the previous line were provided with posters showing their daily constraints and the expected future developments. The objective was to obtain a full assessment from these final users, incorporating the aspects of ergonomics, quality, safety and performance, with a particular emphasis on what could be avoided or improved.
The positive points and those areas requiring improvement, clearly indicated by staff, were integrated into the consultation specifications submitted to different suppliers.
The choice of constructor: partnership
The assessment of the responses of different suppliers should, in consequence, be based on a clear technological approach, meeting the expression of these user needs, and as far as possible offering a combination of technological innovations that could potentially meet the requirement, or proposing an alternative capable of development. Of course, the technical proposal is crucial, as is the commercial offer. However, the capacity of the manufacturer to offer a high quality after-sales service, to offer improvements and to be a true partner, must also be taken into account. The key to a successfully completed project rests in the partnership that must be established between the constructor and the user.
A unique model for all steps of design monitoring
To refine and monitor the project, a single follow-up document is developed in spreadsheet format, reiterating all the points of the specifications. It is used as a document to record and assess the match between the model offered by the supplier and the initial needs determined, at the different stages of design review:
• Specifications, expression of user needs
• Design review
• Design qualification
• Establishment of a traceability matrix for the IQ/OQ qualification tests.
Design/State-of-the-art
In the specific case of the design of an isolator, several critical elements must mandatorily be reviewed:
• The ventilation and air treatment system
• The VPHP decontamination system
• The transfer equipment
• The manipulation equipment
However, in our project, very particular and unusual needs were expressed to the constructor, such as cameras in the isolator, a waste disposal port integrated into the work area and laser scanners to replace the conventional light curtains.
These are all issues for which the isolator constructor must provide responses and clear and innovative proposals.
Regarding this new filling line isolator, here are some examples of responses provided by the constructor relating to the 4 essential areas cited above:
• a new ventilation system design associated with an optimised VPHP system
• a rapid transfer airlock
• a ergonomic glove port design
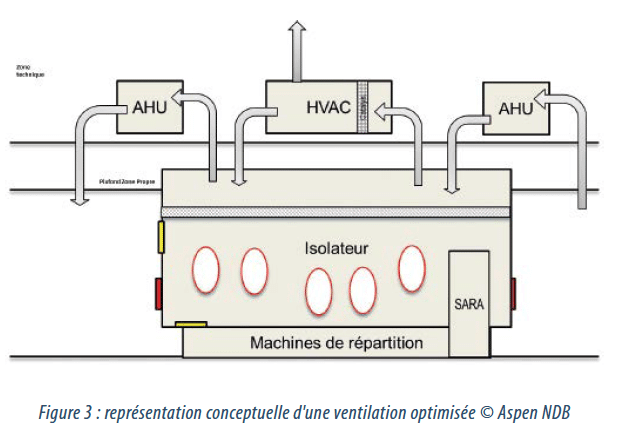
The new ventilation design incorporates:
• the integration of an air distribution plenum with better circulation allowing optimisation of the VPHP cycle,
• a global air treatment system in 3 separate parts. In addition, the air treated by the isolator ventilation system is removed and restored to the surrounding room, enabling substantial economies to be made in the treatment of this room air.
• the integration of a catalytic convertor unit enabling a short VPHP cycle time to be obtained without having to operate air extraction or fresh air intake systems for an additional dilution effect.
The implementation of a bio-decontamination transfer airlock necessitated some technical adjustments for the purpose of complete integration, targeting principally the accessibility of the filling equipment technical area, for better conditions for maintenance operations.
The improvements provided by transparent glove ports gives better visual access, crucial for the control of processes and all manipulations in the isolator chamber. It is in any event, an example of a standard issue usually described as minor, but which is a real plus for the operator with easier manipulation, and therefore safer for the environment and the product.
Mock-up: with the final user
How to ensure that all the elements required or proposed are compatible with the work space. Are the solutions proposed robust? It is essential to carry out a full-size simulation exercise.
At this stage, we are already very familiar with the robots and associated equipment in the workspace, their placement, their technical characteristics, the manufacturing and testing procedures that must be carried out in the isolator chamber, what enters and leaves the isolator.
The full-scale simulation prototype is constructed in wood or cardboard from an actual dimensional drawing of the equipment, and includes wooden models of the robots occupying space inside the chamber.
The advantage of a full scale cardboard model is to allow rapid and precise corrections to be made. The integration of a waste removal port, mentioned earlier, in the work area, was checked and confirmed after cutting a corner off the isolator in a few minutes. The transfer elements (RTP), the manipulation elements (glove ports, suspension bars), and the instruments are positioned in their final places.
But the real added value of the model is to let the actual future users of the line perform manipulations so defining/visualising how operations must be conducted in order to minimise the risks of contamination of the environment or the products, while adding some ergonomic benefits,
Design review: a green light for manufacturing
Here we are at the design review, the prelude to design qualification. At this stage, all the elements have been finalised:
• Drawings, P&ID and plans, 3D models,
• Operational and mechanical specifications,
• Interfaces with other installations
• Elements determined during the mock-up
At this stage, the details are integrated and it is possible to check the match between our need and the final proposed solution. All elements must be described with precision and compared with the initial needs of the users. The detailed plans and diagrams, and the P&ID are subsequently shared and provide a sufficiently substantial level of understanding of the process to begin work on commissioning and on the qualification protocols.
Finally, the design review represents the key stage which enables the initiation of isolator manufacture to be formally authorised.
Manufacture and Assembly: monitoring
From this stage, we enter into the manufacturing phase. However, all the areas for improvement are not determined, and this will apply until the very end of the project. Inspection visits must be carried out, not only to check the project progress, but its quality, and to make the necessary adjustments.
Particular attention must be paid to details and finishing. Inspection visits are also used to obtain an assurance that the equipment will be first tested by the constructor before the first commissioning stage represented by the FAT.
Integration of specific needs: Applications example Laser scanner
Scanner laser
Laser scanners offer users a guaranteed high level of safety. They free up enough work space to facilitate manipulation. And as everyone knows, this also means less risk for the environment and the product.
In our case, 2 types of scanners were used;
The first uses vertical scanning and provides safety of access to the isolator chamber by stopping mechanical movements during production phases. The second uses horizontal scanning and frees up space for the glove extenders during bio-decontamination phases.
Waste removal port
The waste disposal port is entirely integrated in the work area.
A mobile disposal duct, linking the bowl of stoppers to this RTP port was added in order to enable easy emptying and facilitated production line cleaning, in particular without direct manipulation by the operator.
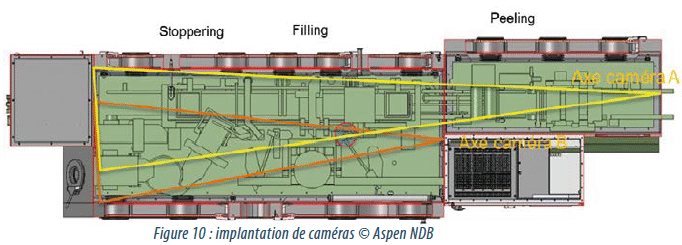
Cameras
Cameras are used to check incursions in the isolator chamber mainly for media fills. The data (images) are recorded and stored digitally. These cameras can also be used in a facilitated diagnosis routine.
Conclusion
Designing equipment in close partnership with the manufacturer throughout the process of the project is the key rule for construction in line with the needs of users and their optimisations.
The important point in the design of an production isolator is not to lose sight of its main objective: to protect the product and the environment in which it is produced. All the work carried out must be focused on the improvements which allow simple, easy use by the manipulators and facilitated, controlled operations. No need expressed must be ruled out immediately.
Each issue must be thought through in partnership with the constructor in order to provide a solution or satisfactory alternative.
Today, our syringe filling line is operational and in full swing. It fully contributes to meeting current market needs and enables the Aspen Notre-Dame-de-Bondeville site to offer a production capacity with the highest standard of quality that is open to new markets and customers.
Some users of this new line who worked on the old line, took part in this project from the mock-up phase.
Many others have arrived very recently. All recognise the improvements made and can express their complete satisfaction.
What better proof of a successful design than the satisfaction and recognition of the final user?

Lionel QUINTON – ASPEN
lquinton@fr.aspenpharma.com
Share article
Glossary
P&ID : Process & Instrumentation Diagram
FAT : Factory Acceptance Test
RTP : Rapid Transfer Port
VPHP : Vapor-Phase Hydrogen Peroxide
QI/QO : Qualification à l’Installation/ Qualification Opérationnelle