Sommaire
- Pourquoi et comment faire une caractérisation thermique d’un produit avant lyophilisation ?
- Les clés d'une qualification réussie
- Comment mener une analyse de risque contamination sur un procédé de lyophilisation ?
- CIP evolution in freeze dryers. Optimize turnaround time & water consumption
- Basiques en simulation de procédés aseptiques et lyophilisation
- Silicon oil detection in pharmaceutical freeze dryer by mass spectrometry
- Les spectromètres de masse. Retour d'expérience
- EN 17272. Nouvelle norme relative à la désinfection par voie aérienne automatisée
En 17272. Tout ce que vous devez savoir sur la nouvelle norme relative à la désinfection par voie aérienne automatisée.
La nouvelle norme européenne EN 17272 est le référentiel de tests à utiliser par les fournisseurs de systèmes de désinfection de surfaces par voie aérienne automatisée pour revendiquer une activité antimicrobienne définie. La norme est basée sur la norme française NFT 72-281, mais contient des exigences d’essai supplémentaires telles que l’homogénéité de la distribution du biocide, le temps de contact global de la désinfection par voie aérienne, les micro- organismes spécifiques utilisés et les volumes d’essai de l’enceinte.

Ce document traite du contexte de l’élaboration de la norme et fournit des directives sur son application.
Un système de désinfection de surface par voie aérienne automatisée permet de diffuser un produit à l’état gazeux, par vapeur ou par air comprimé à partir d’un appareil utilisé hors présence humaine. Les technologies les plus couramment utilisées sont les systèmes de désinfection de surfaces par vaporisation ou par nébulisation de peroxyde d’hydrogène ainsi que les appareils de fumigation de formaldéhyde. Cependant, la norme s’applique également à des technologies moins connues telles que les brumisateurs à libération totale (des conteneurs de produit placés dans une enceinte et activés pour libérer le produit) ainsi que les systèmes de désinfection de surface à l’ozone.
En France, les technologies de désinfection de surface par voie aérienne sont soumises à des normes depuis de nombreuses années. La norme NFT 72-281 a été initialement développée et introduite dans les années 1980 et a ensuite été mise à jour plusieurs fois jusqu’à sa dernière révision en 2014.
En 2010, le Comité Européen de Normalisation (CEN) a jugé utile de créer une norme européenne afin d’évaluer les allégations d’efficacité des nombreux systèmes de désinfection par voie aérienne automatisée disponibles sur le marché. Les systèmes concernés visent à désinfecter les surfaces des installations pharmaceutiques ainsi que les zones sensibles des centres hospitaliers. En s’appuyant sur la norme française NFT 72-281 existante et acceptée par l’industrie, un groupe d’experts dirigé par la France a élaboré une nouvelle norme – la norme EN 17272 – pendant plusieurs années. La norme est issue d’un groupe de travail qui énonce les exigences à l’appui des réclamations dans les domaines médicaux, vétérinaires, industriels et agroalimentaires. Bien que fortement basée sur la norme NFT 72-281:2014, la nouvelle norme vise à répondre à certaines des limites identifiées dans la norme NFT 72-281 et à clarifier certains critères tels que le volume d’essai de l’enceinte.
Quelles sont les quatre principales différences entre les normes en 17272 et NFT 72-281
1. Test d’homogénéité de la distribution
La norme NFT 72-281:2014 exige que l’indicateur biologique soit placé à une distance définie du système de désinfection par voie aérienne automatisée par rapport au volume de l’enceinte dans lequel les essais sont effectués. Les indicateurs sont positionnés verticalement, à une hauteur comprise entre 1,0m et 1,5m du sol, face à l’appareil. Bien que cette configuration indique que le désinfectant et sa technologie de distribution sont capables d’atteindre un certain niveau de réduction microbiologique devant l’appareil à une hauteur de 1,0 à 1,5 m, il ne fournit aucune confirmation que le même niveau de réduction microbiologique soit atteint derrière l’appareil, ou dans les coins de l’enceinte, ou sur des surfaces qui peuvent être horizontales, en particulier celles qui sont orientées vers le sol. La plupart des systèmes de désinfection par voie aérienne automatisée revendiquent une action de la décontamination sur toutes les surfaces exposées ou sur la pièce dans son intégralité. Le critère de la norme NFT 72-281 ne permettait pas d’évaluer une telle allégation. Un test de distribution a été intégré à la norme EN 17272 afin d’évaluer l’homogénéité de la désinfection par voie aérienne automatisée en tout point sur toutes les surfaces de l’enceinte et dans des conditions où les effets de la gravité ou de distribution réduite peuvent affecter le processus. Au total, 8 indicateurs biologiques contenant chacun > 1 x 106 UFC de Staphylococcus aureus sont positionnés dans les angles de l’enceinte. Deux indicateurs sont situés dans un angle bas de l’enceinte – l’un est orienté vers le mur et l’autre vers le plafond.
Deux autres indicateurs se trouvent dans l’angle haut de l’enceinte, diagonalement opposé aux deux premiers, l’un faisant face au plafond et l’autre au mur. Deux indicateurs se trouvent dans l’angle haut de l’enceinte, en face de ceux situés dans les coins supérieurs. Un indicateur fait face au sol et l’autre face au mur. Les deux derniers indicateurs sont situés dans l’angle bas de l’enceinte, diagonalement opposé à ceux situés à l’autre angle bas – encore une fois, l’un face au sol et l’autre face au mur. La disposition des indicateurs est représentée dans le graphique de la Figure 1. Distance des angles = 50 cm (15 cm pour les petites enceintes).
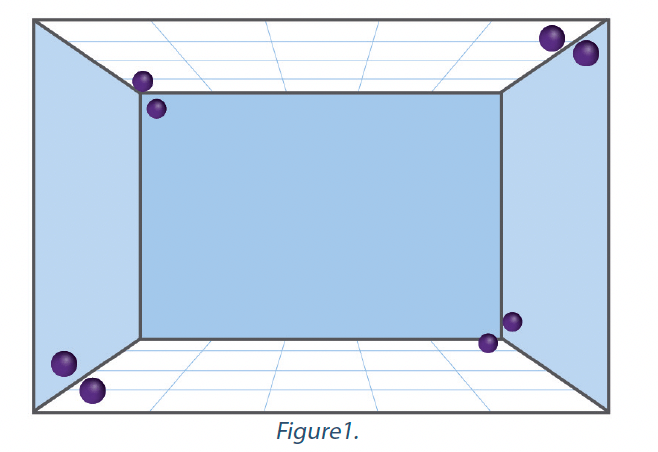
Le système de désinfection par voie aérienne automatisée doit atteindre une réduction de plus de 5-log sur chacun des huit indicateurs biologiques. Si la réduction logarithmique requise n’est pas atteinte sur tous les indicateurs, le système ne peut prétendre passer la norme. Le cycle utilisé pour effectuer l’essai de distribution doit être identique au cycle utilisé dans l’essai d’efficacité de base et le dispositif d’application doit rester au même endroit dans l’enceinte de l’essai.
2. Micro-organismes
Les micro-organismes d’essai dans la norme EN 17272 sont sensiblement similaires à ceux de la norme NFT 72-281 et aux normes d’efficacité du désinfectant EN. Les micro-organismes spécifiques utilisés dans l’essai dépendent des allégations du fabricant concernant l’utilisation de la technologie. Pour revendiquer la conformité à la norme, un fabricant doit, au minimum, satisfaire aux exigences relatives aux essais bactéricides, levuricides et à la distribution. Si un fabricant souhaite appuyer une allégation, par exemple, d’activité contre des virus en milieu hospitalier, il devra satisfaire aux exigences bactéricides, levuricides et virucides pour le domaine médical en plus du test de distribution. Le tableau 2 présente les micro-organismes d’essai et les réductions logarithmiques requises à l’appui d’une allégation de conformité.
Des micro-organismes d’essais spécifiques s’appliquent pour les différentes zones d’utilisation et bien que les exigences de réduction logarithmique soient généralement les mêmes pour tous les domaines d’utilisation, une allégation d’activité sporicide nécessite une réduction logarithmique de 4 dans le domaine médical. L’efficacité contre le Murine Norovirus et l’Adenovirus Type 5 est nécessaire pour soutenir une allégation d’activité virucide dans les domaines médicaux et industriels, tandis que le petit virus non-enveloppé Porcine parvovirus est nécessaire pour soutenir une allégation virucide dans le domaine vétérinaire. L’efficacité contre Acinetobacter baumanii doit être établie dans le cadre des essais pour appuyer une allégation d’activité bactéricide dans le domaine médical.
3. Volume de l’enceinte
La norme NFT 72-281:2014 était peu précise concernant le volume des enceintes dans lesquelles les essais devaient être effectués. La norme indiquait qu’un volume de 30 m3 à 150 m3 devait être utilisé pour établir une allégation d’efficacité mais elle fournissait également des informations sur l’essai de petites enceintes jusqu’à 10 m3. Les exigences en matière d’essai relatives aux petites enceintes n’étaient pas claires et 10 m3 n’étaient pas représentatives des petites enceintes tels que les postes de sécurité microbiologique, les isolateurs, les sas de transfert, etc. La norme exigeait également au fabricant d’effectuer des essais dans une grande enceinte supérieure à 150m3. Cette exigence était impraticable, irréaliste et ne fournit pas de renseignements utiles. La réalisation d’un test selon la norme NFT 72-281 dans une grande enceinte vide n’évalue pas l’efficacité de la décontamination dans l’enceinte mais l’évalue uniquement à l’emplacement des indicateurs biologiques devant le générateur. Les résultats obtenus sont applicables à l’enceinte testée dans les conditions environnementales au début de l’essai. Ils peuvent ne pas être transférables à une autre enceinte de volume égal, mais avec une configuration interne, des matériaux de construction et un rapport surface-volume différents. Il y a peu ou pas de valeur à l’essai.
La norme EN 17272 améliore la clarté des exigences d’essai par rapport au volume de l’enceinte. Concernant les enceintes de petits volumes, les fabricants doivent effectuer les tests dans une enceinte d’un volume compris entre 0,25 m3 et 4,0 m3. Pour les revendications d’utilisation dans des enceintes supérieures à 4 m3, les fabricants doivent utiliser une salle dont le volume est compris entre 30 m3 à 150 m3. L’exigence peu pratique et inestimable de tester dans de grandes enceintes de plus de 150 m3 a été supprimée.

La norme EN 17272 reconnaît que l’essai dans une enceinte vide n’est pas équivalent à l’essai dans une salle de volume identique contenant une charge (poste de travail, équipement, etc.). La norme indique clairement que son objectif est de fournir un référentiel de tests permettant de confirmer l’efficacité d’un système de désinfection par voie aérienne automatisée. La norme identifie que chaque cycle de décontamination est unique et que les cycles doivent être validés en pratique à l’aide d’indicateurs biologiques appropriés ou d’indicateurs chimiques étalonnés. Cette exigence appuie la position de l’Agence européenne des produits chimiques (ECHA) détaillée dans la section 8 de l’accord technique sur l’efficacité des biocides.
4. Temps de contact global de la désinfection par voie aérienne
Un cycle de désinfection par voie aérienne automatisée se compose de 4 phases :
Conditionnement – Préparation et conditionnement du système de désinfection par voie aérienne automatisée et de l’enceinte à décontaminer en vue de l’introduction du gaz, de la vapeur ou de la nébulisation.
Diffusion – Introduction du gaz, de la vapeur ou de la nébulisation Contact – L’agent désinfectant reste en suspension sur les surfaces de l’enceinte.
Aération – Élimination du désinfectant afin de réutiliser l’enceinte.
La norme NFT 72-281 prenait en compte uniquement le temps de contact et non le temps global pendant lequel le désinfectant a été en contact avec les surfaces de l’enceinte. Par exemple, le système A pourrait injecter 20 g/m3 de désinfectant sur une période d’une heure et avoir un temps de contact de 10 minutes, et le système B pourrait injecter 20 g/m3 sur une période de 15 minutes et avoir un temps de contact de 10 minutes. Les cycles et les temps de contact réels associés avec la surface sont très différents, mais les temps de contact signalés sont identiques à 10 minutes. La norme EN 17272 introduit le concept de temps de contact global de la désinfection par voie aérienne pour représenter plus précisément le temps de contact global de l’agent désinfectant. Cette période débute lors de la diffusion du désinfectant dans l’enceinte et se termine au début de l’aération.
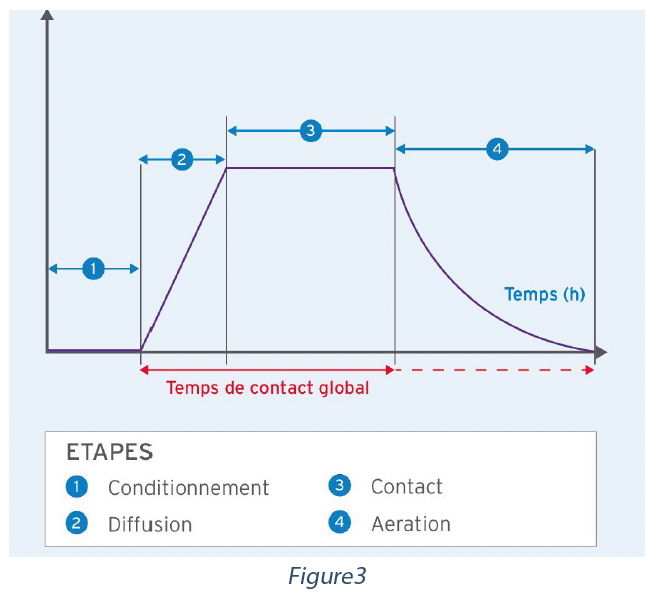
La norme NFT 72-281 prenait en compte uniquement le temps de contact et non le temps global pendant lequel le désinfectant a été en contact avec les surfaces de l’enceinte. Par exemple, le système A pourrait injecter 20 g/m3 de désinfectant sur une période d’une heure et avoir un temps de contact de 10 minutes, et le système B pourrait injecter 20 g/m3 sur une période de 15 minutes et avoir un temps de contact de 10 minutes. Les cycles et les temps de contact réels associés avec la surface sont très différents, mais les temps de contact signalés sont identiques à 10 minutes. La norme EN 17272 introduit le concept de temps de contact global de la désinfection par voie aérienne pour représenter plus précisément le temps de contact global de l’agent désinfectant. Cette période débute lors de la diffusion du désinfectant dans l’enceinte et se termine au début de l’aération.
Si aucun processus d’aération n’est utilisé, cette période se termine lors de la récupération des indicateurs biologiques. Si l’aération est utilisée, les détails du taux d’aération doivent être inclus dans le rapport d’essai.
5. Rapports de la norme EN 17272
La norme EN 17272 fournit des instructions sur les informations à inclure dans le rapport d’un essai et comprend :
– Identité du laboratoire d’essai
– Nom du produit (désinfectant) et de l’appareil à l‘essai
– Type de distribution (vaporisation, brumisation, nébulisation, atomisation, etc.)
– Numéro de lot du produit désinfectant
– Numéro de série de l’appareil
– Type d’indicateur biologique utilisé
– Distance entre l’appareil et les indicateurs
– Schéma à l’échelle de la configuration du test
– Temps de contact global
– Temps de diffusion
– Temps de contact
– Temps d’aération
– Taux d’aération (ou taux de renouvellement d’air)
– Quantité de désinfectant utilisé
– Volume exact de l’enceinte de test.
Le rapport d’essai doit indiquer le ou les groupes de micro-organismes qui ont fait l’objet d’essais et indiquer si les résultats des essais sont “conformes” ou “non conformes” aux exigences de l’essai. Pour l’essai de distribution, une réduction de 5-log doit être obtenue pour chaque indicateur biologique.
Les organismes de réglementation et les acquéreurs de systèmes de désinfection par voie aérienne automatisée devraient demander des copies du rapport d’essai EN 17272 du fabricant et en examinant les données d’essai pour déterminer les conditions dans lesquelles le système a été testé. Les fabricants peuvent revendiquer la conformité à la norme EN 17272 sur la base de tests contre les bactéries végétatives et la levure. Les fabricants qui cherchent un système pour traiter les spores (comme Clostridioides difficile), les champignons ou les virus (comme le SARS CoV2) devraient s’assurer que le système a été testé par rapport aux micro-organismes d’essai pertinents pour appuyer ces allégations. Les utilisateurs devraient évaluer les allégations des fabricants relatifs au temps de contact global, car les fabricants peuvent uniquement déclarer le temps de diffusion du produit (parfois appelé temps d’application) ou le temps de contact, ce qui peut entraîner une sous-estimation de la durée globale du cycle du système.
Conclusion
La norme EN 17272 structure et améliore les fondements élaborés par la NFT 72-281. Le document d’orientation sur l’efficacité PT 1-5 du règlement européen sur les produits biocides (BPR) identifie l’utilisation de la norme européenne dans l’évaluation des applications de produits biocides impliquant des systèmes de désinfection par voie aérienne automatisée.
Le système Bioquell de bio-décontamination par vapeur de peroxyde d’hydrogène a été testé dans des petites et grandes enceintes selon la norme EN 17272, pour les domaines médicaux, vétérinaires, alimentaires et industriels et soutient les allégations relatives à l’activité bactéricide, mycobactéricide, sporicide, fongicide, levuricide et virucide. Une gamme d’indicateurs biologiques et chimiques calibrés est proposée, qui peuvent être utilisés pour vérifier la performance du cycle de désinfection par voie aérienne tel que requis dans la norme EN 17272 et dans l’accord technique d’efficacité sur les biocides de l’ECHA.
Partager l’article

John CHEWINS – ECOLAB
john.chewins@ecolab.com




