Summary
- Injectable Combination Products. Issues and challenges for industry.
- Deadline Extension or Not: Key Points for Class I Manufacturers
- The new challenges of injectable medicines
- Clinical Investigation of Combined Medical Devices Under New Regulation (EU) 2017/745 (MDR)
- Contribution of the physicochemical characterization of the materials constituting medical devices for rationalisation of their biological assessment.
- Steering Cleaning Validation Performance : A Key Industrial Challenge.
- Cleaning Process Validation: Why and How to Validate Analytical Methods and Related Sampling Methods
- A Risk-Based Approach to Stainless Steel Equipment Maintenance in cGMP Manufacturing Environment
- Single Use Systems vs Re-Usable Stainless-Steel Equipment. Compliance & Quality Perspective.
The new challenges of injectable medicines
The past few years have seen a tremendous development of injectable drugs with devices that allow patients to self-administer their therapy in an easier, safer and freer way. They no longer need to go to their doctor or hospital as often and become more independent and flexible actors in managing their condition. These new therapies, the emergence of new technologies and the arrival of a new European Medical Device Regulation (EU) 2017/745 MDR are causing a booming of the pharma sector!
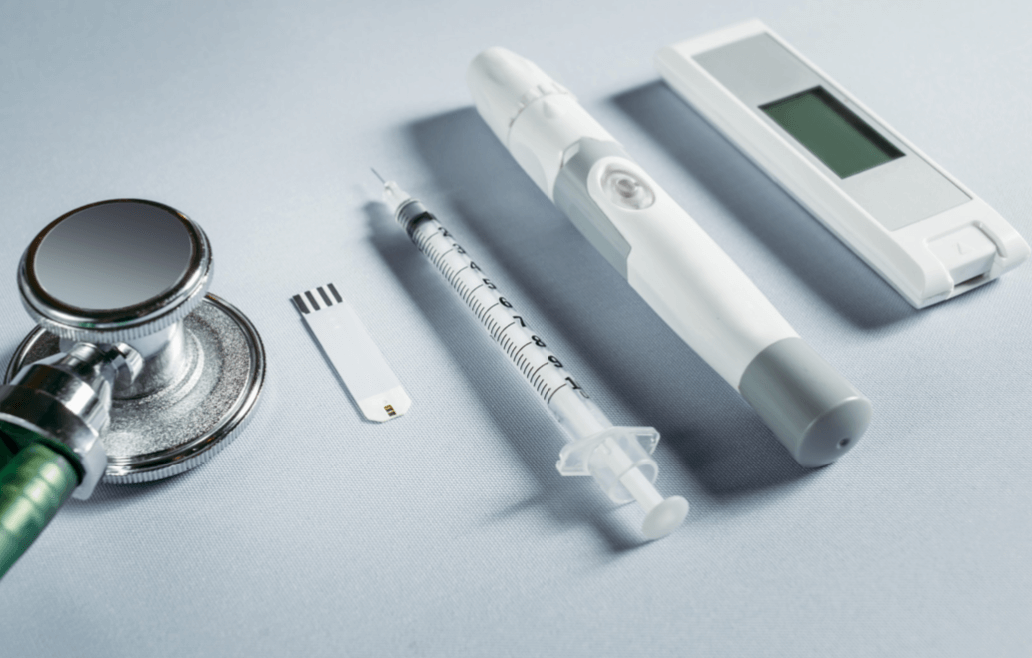
Many new injectable drugs reached the market in the last decade, whether original preparations or generics, to treat an increasing number of patients suffering from chronic diseases, cancers or autoimmune diseases. Parenteral injections enables the development and assimilation of complex and more viscous molecules which provide more effective solutions for the treatment of these diseases. In fact, injections do not pass through the gastrointestinal system like oral medicines but go directly into the blood system and therefore the bioassimilation is enhanced.
In an effort to reduce the costs of treatment and care, many of these new pharmaceutical products are being developed for subcutaneous injection by the patient at home rather than by a healthcare provider in a medical environment. This trend initially comes from diabetes treatments. Indeed, many injection devices were first developed for the administration of insulin and were later used in other therapeutic areas.
Nowadays a self-injecting patient expects to receive an injection pen or a wearable on-body injector rather than a syringe-vial kit! Therefore many ready-to-use subcutaneous injection devices have recently emerged to support patients. In developed countries, these devices are mainly distributed in a single ready-to-use unit to facilitate handling by the patient, which also means single use. In fact, insulin pens in Europe and the United States are mainly disposable single-use products, while in Asia it is very common for patients to receive reusable pens.
1. The rise of a new European regulation
This trend has also been followed by European legislators in issuing a new Medical Devices Regulation (EU 2017/745 MDR) which will come into force in 2021 (postponed from 2020 to 2021 by the European Commission due to the Covid-19 pandemic). It replaces the Medical Devices Directive 93/42 / EEC and aims to align EU legislation on technical progress, on developments in medical science and on the legislation of other countries (especially the United States through FDA guidelines). Moreover, the MDR specifically addresses the requirements for products combining a drug and a medical device in a single entity unit through article 117, the so-called combination product.
These combination products have been regulated so far as medicinal products without specific examination of the injection device constituent. And this is the tour de force of the European legislators who indirectly amend the Directive 2001/83/EC related to medicinal products for human use through another distinct regulation (MDR, article 117). Indeed, from the entry into force of the MDR, it will become mandatory to provide either an EU declaration of conformity or an opinion from a Notified Body on the device constituent for registering a new combination product. And this will bring lots of new challenges for the pharmaceutical companies.
This means that pharmaceutical companies will have to submit complete technical documentation for the injection device to a Notified Body, if this device does not bear a declaration of conformity. This Notified Body will then make an in-depth review of this technical documentation and issue a positive/negative opinion on the compliance with the requirements of the MDR (Annex I of the MDR). This opinion will then be submitted by the pharmaceutical company to the European Medicine Agency (“EMA”) as part of the registration dossier for the final product.
There will be many challenges in the future for pharma companies who develop combination products
- The injection device can no longer be underestimated. Up to now in many cases it had been considered, at most, as secondary packaging. Development timelines and budgets will dramatically increase. Indeed, it will become necessary to have dedicated resources, competences and especially processes in place (or a QMS) for medical devices in order to prepare the technical documentation mainly comprising technical and functional requirements, a detailed technical description of the injection device, design verification reports, a risk management file, a design validation through usability and human factors and potentially a clinical evaluation report.
- In addition, it will also be essential to collaborate with a Notified Body certified for the new MDR. And even though the entry into force was postponed to 2021, this could turn out to be a bottleneck in the whole process because currently very few Notified Bodies are certified under MDR and only a part of them offer to provide an opinion according to article 117.
- The change induced by the MDR must also be integrated by the device platforms vendors who may still be reluctant to share the necessary information for a submission to a Notified Body by the pharmaceutical company. This aspect has to be taken into account in the negotiation phase and in the selection of a competent, well-prepared and cooperative vendor.
- And at the end of the day, the responsibility for these devices on the market lies with the pharmaceutical companies. The new MDR defines a number of specific requirements once the product is launched (Post Market Surveillance / PMS) which obliges pharmaceutical companies to have a proactive approach in the management and in the regular assessment of their medical devices.
2. The American influence
This new MDR is aimed to fill a gap in the regulation of combination products compared to some other regions, mainly the United States. Actually, the FDA has released combination products directives and guidance for a long time. The European Union is now integrating this concept in its regulation while raising the bar for all medical devices at the same time. Indeed the scandals of the early 2010s, where suppliers voluntarily defrauded the quality of medical products and endangered their patients, triggered the European Union to increase its requirements and update its regulations through this new MDR.
But more stringent European regulations than the American ones could also curb innovation in small European structures and reduce the portfolio of marketed products in Europe. It will become more difficult to launch medical products in Europe than in the United States.
Indeed, in order to submit a new medical device in the United States it is possible to use elements of the technical file from an equivalent marketed medical device. But following the implementation of the MDR, it will now become mandatory to provide a comprehensive file containing all the clinical data for any new medical device in Europe. For a new combination product, it will first be necessary to obtain the positive opinion of a Notified Body for the medical device and then the complete registration dossier of the drug will have to be reviewed by the EMA, which will lengthen the registration period for these products in Europe. The requirements for economic operators (such as importers or distributors) are also much more detailed in Europe than in the United States. Even the large companies would probably prune their catalog of marketed products in Europe and the United States will de facto strengthen their position as a target market. Europe will then come after in the commercial strategy. On the other hand, in the specific case of combination products, these new requirements concerning the technical file will put European companies on track for a registration of their product in the United States. Indeed, it will become easy to compile a registration file for the FDA based on the European one.
3. Innovation as a differentiation strategy
But the development and marketing of injectable drugs will not go down. The expiration of patents for numerous molecules opens the door to many new generics or biosimilars and the demographic evolution of a sedentary and aging population witnesses the increase of chronic diseases and cancers, which require new therapeutic responses.
The launch of many generics or biosimilars exposes pharmaceutical companies to fierce competition when patents on a molecule expire. Given the limited possibilities to differentiate their product from competitors with the same molecule, pharma companies are now trying to use the injection device as a strategic differentiation asset. If a new market entrant offers a more attractive and more user-friendly product for the patient, it will have a strong competitive advantage without having to fight on the pricing policy. This leads to the development of more differentiated injection devices in terms of outer design and value proposition.
Amgen is a case study of a device differentiation strategy in order to defend their market position (Neupogen / Neulasta) facing the emergence of biosimilars. Initially launched in a pre-filled syringe configuration, Amgen’s Neulasta then evolved as a wearable on-body injector for improving the patient convenience and reduce the patient handling effort by medical personnel. There are many similar examples of products whose administration device evolves over time while the drug formulation remains unchanged. A medicine initially available in a vial is then marketed in pre-filled syringes in the next version. This pre-filled syringe then evolves into an autoinjector (injection pen) to make it even easier for the patient to self-inject at home.
4. Connected devices and digital solutions
Today we witness different streams of innovation strategies for future products. One major trend is connecting these injection devices to smartphones to bring new functionalities helping the patient. Another strategy is to space out the doses frequency in chronic disease to relieve the patients from their condition. In addition, payers and health insurers are increasing pressure to reduce costs leading all the health stakeholders to seek solutions to empower the patient and limit the care in a medical environment.
Nowadays, all household devices are launched with a connected app. Whether it is a kitchen appliance, a car or a treadmill, it can be managed and tracked on a smartphone, showing trend graphs, sending reminder notifications or even allowing remote control. Injection medical devices are no exception. There are already insulin pens that inform patients about their last dose through a connected app, remind them of their next injection and show them a trend graph of the last weeks. And this type of features will also be ruled by the MDR. Developing a medical app or software ultimately requires the same skills used for a medical device.
When defining the strategy for a new pharmaceutical product, it still remains important to take into account the challenges and disadvantages associated with this type of connected solutions, such as:
- The intrusive aspect of these technologies for patients who do not want to be constantly confronted with their condition.
- The age of usually elderly patients who are not comfortable with this kind of technology.
- The business model which must be clearly defined. It is not easy to turn a software or a medical application into cashflow. This feature can then be considered as an add-on bringing added value to a pharmaceutical product.
- The complexity for a pharmaceutical company to develop this type of solution. These digital products bring new objectives as well as new stakeholders (IT, cloud computing, UI / UX design, etc.) who are not necessarily familiar with the requirements of the pharmaceutical and medical industry.
- The management of the generated data, the data security and the confidentiality (GDPR) as well as the registration of these new solutions which, according to their features, will be considered as medical devices. Therefore, it will be mandatory to compile a registration dossier including a design validation through a summative human factors study as well as a clinical evaluation report in order to comply again with the new MDR.
Despite these challenges related to connected devices, no pharma company wants to miss this new digitalization era that brings a lot of possibilities for patients and opportunities for the entire health system.
Indeed, this empowering approach for the patient brings a solution which goes beyond the medication and which supports the patient throughout his whole care journey.
These tools ultimately enable much more rigorous patient adherence to the therapy, which is also a strong argument for biotechnology companies with very high value products in their negotiation with payers.
5. Spacing out the injections
Offering drugs which require less frequent injections is also an approach to differentiate from the competition. As an example, a patient with a chronic disease should no longer be administered a daily or weekly injection, but only once a month. This type of long-acting or controlled-release treatment often requires coupling the active molecule to a carrier, which has the effect of increasing the size of the molecules and therefore the viscosity of the drug liquid. In other cases, the deliverable volume will be increased in order to provide enough drug substance for the entire period between two doses.
Larger, denser or more viscous injections also mean new requirements for injection devices such as a more powerful activation mechanism or larger primary containers. Nowadays, a lot of development projects use electromechanical injection devices (like wearable infusion pump) to allow the controlled delivery of large volumes over longer injection times.
6. Ally with reliable partners
In order to successfully carry out these cutting-edge development projects in an efficient way, it becomes mandatory for the pharmaceutical and medical companies to find out new solutions. The injection devices are becoming more complex and the regulatory requirements are increasing therefore the challenges are numerous:
- Targeting precisely the unmet user needs of users and patients and defining precisely the objectives to meet them.
- Implementing more agile and faster development processes into a traditional stage-gate approach. Minimum Viable Product (MVP) and agile project management can be integrated into a quality system to develop digital solutions. These iterative methods allow stepwise development phases and repetition of end-user formative tests all along the development process.
- Selecting and using more efficient tools for project documentation and complex requirements traceability. Indeed, each technical or functional requirement must be verified or validated during the project while making the link with risk management. Modern tools exist to facilitate structuring these requirements (e.g. tachysCP).
It is complicated nowadays for many companies to find all these skills and competences internally. Alliances with strategic partners can therefore become crucial. It is often more efficient to collaborate with external partners for punctual complex projects rather than to train or recruit internal staff for a limited number of projects and tasks. But all these challenges should not hide how these new products will improve the lives of patients by facilitating their therapy and offering more effective solutions in the future.
Conclusion
The sector of injectable drugs is today facing new technological and regulatory challenges which also open up fantastic opportunities for patients and the whole healthcare system. Future medicines will treat diseases in a more effective and less restrictive manner. The new regulations will ensure increased levels of efficacy, quality and safety of these medicines. This new turn must also be seen as an opportunity to improve processes and to increase the value proposition for new applications and patient needs. The players who will best adapt will have long term tools to stay at the forefront of a growing pharmaceutical sector, more vital than ever for the health of populations.
Share article

Thierry JOMINI – Anteris Helvetia AG
tj@anteris-helvetia.com
Glossary
Data security : Sécurité des données
EMA : European Medicine Agency. Agence européenne des médicaments
FDA : US Food and Drug Administration. Administration américaine des denrées alimentaires et des médicaments.
GDPR : General Data Protection Regulation. Règlement général de l’UE sur la protection des données
MDD : Directive 93/42/CEE sur les dispositifs médicaux
MDR : Réglementation européenne des Dispositifs Médicaux (UE) 2017/745 MVP : Minimum Viable Product. Produit minimal viable
UE : Union Européenne
UI/UX design : Interface et expérience utilisateur
References
2. Medical Device Directive 93/42/CEE : https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:31993L0042&from=FR
3. Medicinal Product Directive 2001/83/CE : https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:32001L0083&from=FR
4. Draft guideline on quality requirements for Drug-Device combination https://www.ema.europa.eu/en/documents/scientific-guideline/draft-guideline-quality-requirements-drug-device-combinations_en.pdf
7. https://aphadolie.com/2018/11/26/scandale-des-dispositifs-medicaux-les-lacunes-du-controle-des-protheses-medicales-revelees-par-une-enquete-internationale-videos/
9. Biabnext Clipsulin https://www.diabnext.com/fr/clipsulin-c3/
10. https://anteris-medical.com/tachyscp.html




