Sommaire
- La campagne on y gagne ! Le travail en mode campagne, une avancée sur la productivité des isolateurs.
- Inspection visuelle : principaux constats des inspections de l’ANSM
- Validation des nettoyages des équipements de production : Contrôle visuel, habilitation du personnel au « visuellement propre »
- A Refresher on Disinfectant Wet Contact Time
- RDM et RDIV de l’UE : Les produits combinés désormais soumis au même degré de surveillance que les dispositifs médicaux autonomes
- Fiabilité de la lignée monocytaire Mono-Mac-6 pour la détection des pyrogènes endotoxiniques et non-endotoxiniques
- Indicateurs biologiques, pousses aléatoires - votre cycle de décontamination est-il réellement en cause ?
- Determining a Strategy for Container Closure Integrity Testing of Sterile Injectable Products
- Efficient Control Strategy enabled by structured Knowledge
- Une solution performante pour les dénombrements en temps réel des colonies sur membranes dans l’analyse microbiologique avec ScanStation
Efficient Control Strategy enabled by structured Knowledge.
Control Strategy, derived from current product and process understanding, is built-up throughout Pharmaceutical Development, becomes a central repository for Regulatory filing, and the reference to ensure Product Quality and Process Performance during Commercial Manufacturing and Product Lifecycle (OPV, Change Management).

Two enablers ensure the robustness of the Control Strategy: Quality Risk Management and Knowledge Management, supported by regulatory guidelines, particularly ICH Q10. With the increased complexity of the medicinal products and manufacturing processes, and the need to supply innovative therapies more and more rapidly for the patient needs, awareness has raised in the industry to implement jointly and efficiently these two enablers. The central question today is how QRM and KM can be improved to accelerate the products development, the products supply and innovation throughout the lifecycle? Which kind of knowledge should be managed? How to improve the knowledge leverage for an efficient use? What are the solutions to increase the relevant knowledge overtime (i.e. obsolete data removed), thanks to key contributors and Subject Matter Experts?
If the solutions are not totally developed and available, initiatives are emerging in the Health industry to improve knowledge flows, Knowledge Management tools and technologies (through Pharma 4.0 solutions). A mindset evolution in the Knowledge Management will be necessary to achieve the goals, but today, everyone aligns on the need: improve structuring, management, transfer and use of the Knowledge. The availability of the most up-to-date knowledge in real time, structured in databases, and the ability to share this knowledge efficiently through comprehensive and powerful digital tools will surely benefit product innovation, product quality and facilitate product lifecycle management.
1. Introduction
The Control Strategy, ensuring product quality, has always been a requirement, but was historically mainly ensured by end-product testing and narrow control of manufacturing processes and materials without a comprehensive understanding of the relationships between process parameters, material attributes and product quality attributes.The Control Strategy concept has evolved with the implementation of ICH guidelines Q8 to Q121, the 10 past years. A new pharmaceutical paradigm has emerged, with Quality by design approach highlighting science & risk-based approaches. In an enhanced approach to manufacturing process development using Quality by Design (QbD) concepts, an enhanced Control Strategy is based on a better understanding of the product and the process allowing to identify which material attributes and process parameters should be controlled. Scientifically sound and based on risk assessments, this approach allows Control Strategy to focus on fundamental elements influencing the product Critical Quality Attributes and ensuring to meet product quality.
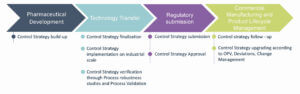
Control Strategy including different types of control to assure product quality, is built during Pharmaceutical Process & Analytical Development and consolidated during Technology Transfer, proposed by the applicant in the Regulatory filing and approved by regulators. Building an efficient Control Strategy for the Commercial Phase of the product, relying on the basics of the Product and Process risks appear even more necessary and relevant as the ICH Q121 has been published to facilitate Change Management during the product lifecycle, encouraging innovation and continuous improvement.
The need for a robust Control Strategy, built in development and managed throughout the product lifecycle raised the need of effective Knowledge Management and Quality Risk Management use throughout this lifecycle.
Knowledge Management and Quality Risk Management have also been recognized by regulators as key enablers, strongly linked to ensure pharmaceutical products quality, providing the means for science- and risk- based decisions related to product quality (ICH Q10).
If there is a consensus that knowledge is critical to successfully manage the product lifecycle, there are significant challenges to ensure that the right knowledge efficiently flows across the product lifecycle to build and manage the Control Strategy.
2. Current state and challenges
Initially developed and implemented during product development for production of clinical trial materials, Control Strategy must be updated for commercial scale production before regulatory dossier submission. Control Strategy can be challenged, reviewed when new product/process knowledge is gained during manufacturing of the commercial batches (communication with Health Authorities for Control Strategy update is determined by the element to be changed).
To develop and manage the Control Strategy, the use of QRM and KM have become compulsory throughout the product lifecycle:
• pharmaceutical development
• technology transfer and process validation
• preparation of the regulatory dossier submission
• commercial manufacturing: deviations and change management processes
2.1 Pharmaceutical Development (Construction of the Control strategy during Pharmaceutical Development)
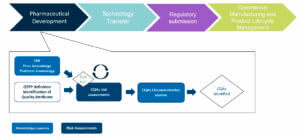
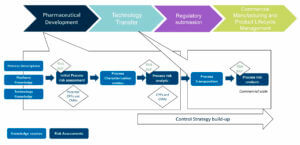
Control Strategy is being constructed during late stages of the pharmaceutical development, is consolidated and finalized during Technology Transfer and implemented at the scale of the commercial manufacturing process. It is followed and verified during Process Validation stages and is an essential part of the CMC regulatory dossier and the basis for Commercial Manufacturing to ensure Product Quality. During the product lifecycle, the Control Strategy can evolve according to process monitoring feedback and Change Management process (Figure 1).
At each stage of the product lifecycle and the Control Strategy build-up, product and process knowledge aided-risk assessments are performed. Using risk-management tools and iterative risk assessments are crucial for developing a control strategy that will be accepted by Health Authorities.
Figure 2 illustrates the different risk assessments performed during product characterization phase to obtain the final list of CQAs.
Figure 3 illustrates the different risk assessments performed during the process characterization phase to identify the Critical Process Parameters and Critical Material Attributes.
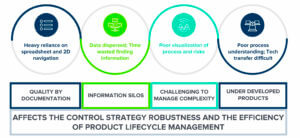
Product and process knowledge is today largely tacit, particularly during early stage of development phase. Collecting a relevant and usable knowledge to perform these risk assessments is often a challenge. People who create knowledge are often separated by their location and the projects time from the knowledge users. Issues in the knowledge flow from early product development through the lifecycle are often encountered. Typical issues that are met are:
• the lack of data formalization,
• the unavailability of the SMEs who participated in the early stages of the project (SMEs do not necessarily stay at a company for the entire duration of a product lifecycle)
• the organizations in silos that slow the communication loops and knowledge transfer between product development teams, process development teams, Regulatory Affairs, industrialization teams
Knowledge loss and tacit knowledge are a central question, but unstructured knowledge is another one. Explicit knowledge is often stored in unstructured repositories files.
Another typical situation encountered is a collection of data assembled at the end of each development gate, leading to large development reports retracing several years of development. Freestyle narrative reports are available, but they are not directly linked to the goal: Product and process relationships (CQAs, CPPs and CMAs identification). Then, a high workload is needed to transform these narrative elements into a comprehensive and structured risk assessment tool.
Knowledge leverage lacks efficiency and at each step of QbD approach, these risk assessments require some efforts. They consume time and resources to collect historical knowledge, compile the data and provide the scientific rationales. These statements are also applicable during Technology Transfer and the preparation of Process Validation.
2.2 Technology Transfer and Process Validation
During Technology Transfer, the process is transposed to the commercial scale and Control Strategy is usually finalized at this stage, requiring another round of process risk analysis focused on industrial scale impacts. This time, the CPPs and CMAs identification has already been performed, the goal is to identify how they are controlled at industrial scale and with the technologies in place in the plant. This part of the Control Strategy is often called Process Control Strategy.
To formalize the Process Control Strategy on the commercial scale process, an update of the process risk analysis is needed. A typical situation when performing the process risk analysis at industrial scale is to redevelop a new risk analysis tool because the one used in Development phase does not comply with specific requirements, SOPs, or templates of the manufacturing site. There is a lack of standard tools and templates for managing knowledge and this situation adversely affects the efficiency of Transfer Technology. There is a need for flexible tools and rules (complexity varying according to the stage of the lifecycle) but also harmonized definitions to avoid misalignment when preparing the regulatory dossier.
In the scope of Knowledge Management, we can observe that successes and failures are not systematically captured across the lifecycle and induce to reperform some experiments by engineering teams that were maybe already experienced by development teams.
Process changes history and the rationale for each change can be difficult to collect unfortunately, yet it is key for the process understanding and the future product lifecycle.
Concerning the tools, Excel spreadsheets are developed to formalize Product & Process risk assessments.. No one can deny that there are some risks of discrepancies between files when redundant information is present and when updates are performed. The cost to maintain these tools is not negligible and has to be taken into consideration when defining the maintenance and update frequency of these Excel Spreadsheets and associated reports.
In the scope of knowledge management, transfer and sharing, the usual Microsoft Office tools (word files, excel sheets,) provide limited visualization solutions. To create instructive visuals, Experts must generate them manually, which is time-consuming and expensive, and can be sources of error as they are manually created.
2.3 Regulatory dossier submission
When comes the time for the regulatory dossier preparation and submission, the worst situation that could happen is that the Regulatory Affairs drafting the CMC sections were not involved during previous stages of the project. Some elements of the Control Strategy described in the dossier will become regulatory binding and the dossier drafting should be performed as a team. The team should consist of the Regulatory Affairs (CMC Managers), product and process SMEs, Projects teams involved in risk assessments, and anticipated in the previous stages of the project.
This teamwork will enable the provision of a relevant overview of Control Strategy and detailed scientific justification and rationales of each individual element which are presented in the Regulatory dossier.
2.4 Commercial manufacturing: deviations and change management processes
During commercial manufacturing, Control Strategy is the key element to ensure product quality and process performance. The relationships between materials and process parameters with the product CQAs (i.e. the critical elements assuring the product quality) must be perfectly understood to efficiently manage the process deviations and the Change Management process. If this is not the case, Deviations management and Change Management are very challenging, time consuming if the scientific rationales and knowledge are not easily accessible.
Many challenges affect the Control Strategy robustness and the efficiency of the Product Lifecycle Management. Nevertheless, awareness emerged in the Pharmaceutical industry to improve the data and knowledge availability to support the Control Strategy.
3. Needs and solution elements
To answer to the current state and challenges described before, pharmaceutical industry needs to work on the effectiveness of the knowledge structure, management, flows and use. Some key elements are summarized (do not represent an exhaustive list):
• Improve the availability of an up-to-date knowledge, at any time of the lifecycle
• Be able to easily update data and the knowledge progression
• Better support the documentation and traceability of process development
• Improve the Knowledge structuring and accessibility
• Improve the capture of platform knowledge and prior knowledge
• Improve the availability of subject matter expertise
• Improve the communication loops
In addition, to gain efficiency in all stages of the lifecycle, from development to commercial production, the following solution elements are intended to decrease the cost, workload and level of effort needed at each stage:
• Develop iterative risk assessment tools using common definitions and wording
• Develop common key methodologies and concepts that will be used in QbD/QRM implementation throughout the product lifecycle
• Facilitate communication loops (to express needs and feedbacks) in the product and process development lifecycle
• Use visualization tools and technologies allowing a more efficient sharing of the knowledge, being more powerful in the risk communication, recommended by guideline ICH Q9.
• Improve the sharing of information about risk and process design between the decision makers and other stakeholders
International working groups like the BPOG2 (BioPhorum Operations Group) already started the work to identify solution elements and has developed a Knowledge mapping for the biopharmaceutical industry in CMC business (Process Development, Control strategy development and Technology Transfer). This group proposed solutions to answer to current state and challenges, around the guiding principles of Knowledge Management considering People, Process, Content and Technology.
The feedback loops in the product and process development lifecycle are a good example of a solution element considering Process (who, when, how), and Content (what). A typical feedback loop that can be used as an example is the relationship between manufacturing and product/process development. To develop a manufacturing process that will answer to the manufacturing needs and constraints, a good practice is to share between Development and Manufacturing teams to gather manufacturing sites experience for process control capability (according to the current technology available). Considering the manufacturing needs and constraints is key to design the appropriate studies reflecting enough process variability and flexibility. On the other side, manufacturing teams should be aware of development choices on essential process design elements to avoid reproducing failures during process transfer, that could already occur in previous stages of the lifecycle. The involvement of Regulatory stakeholders (CMC) during the Development phase is also a best practice, that is not always followed. Later, during commercial manufacturing phase, sharing between Development and Manufacturing teams will support the continuous improvement both for future development and current manufacturing challenges (i.e. deviations handling, change management).
To summarize, the involvement and engagement of cross-functional teams for all QbD steps is crucial for the project success as well as the implementation and use of iterative tools and common methodologies throughout the lifecycle. These principles will enable to collect the scientific rationales and populate the risks assessments on a regular basis and avoid knowledge loss and jerky effects on workload before each project gate.
Considering the “Technology” as a KM guiding principle, Industry needs the availability of appropriate tools to answer to their knowledge flows improvement. Structured databases are needed to support the amount of data that will be generated throughout the lifecycle. An illustration of structuring is shown Figure 5.
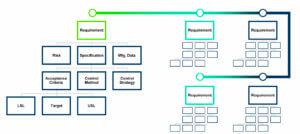
To answer to the drawbacks of Excel Spreadsheets or Word documents to aggregate data from the earliest stages of process design and development through technology transfer and commercialization, IT softwares are currently developed and commercialized, enabling availability of electronic data (easily accessible and searchable) providing an enhanced quality-by-design vision. These softwares rely on structured databases and represent the future of the QbD approach implementation. Why ? They facilitate and accelerate the Knowledge Management, transfer and sharing, thanks to the creation of instructive and powerful visuals, enabled by automated visualization tools.
4. Opportunities and Benefits
As developed throughout this article, the benefits of improving the knowledge flows and structure to support iterative risk assessments and new knowledge inputs during Development, Submission and Commercial Phases are obvious and abundant to increase the efficiency of pharmaceutical companies, such as:
• Accelerate time to market and decrease the cost of drug development
• Accelerate decisions, based on better information
• Accelerate the Technology transfer phase
• Reduce the workload and risks for Regulatory dossier submission and approval
• Accelerate the deviations handling and troubleshooting during commercial phase
• Enable a more efficient Change Management process as proposed in ICH Q12 guideline
As regards to ICH Q12, the aim of this guideline, published in final version (Step 4) in November 2019, and currently in Step 5 (implementation phase) is to provide a framework to facilitate the management of post-approval CMC changes in a more predictable and efficient manner. The implementation of this approach is:
• on one side, a huge opportunity to provide more flexible regulatory approaches, reduce the number of regulatory variations and simplify the communication process (i.e. reporting categories for changes) between HA and MAH, reducing the regulatory burden for the pharmaceutical companies
• on the other side, subject to increased product and process knowledge, to the use of science- and risk-based approaches in drug development and regulatory decisions (supported by effective Pharmaceutical Quality System).
The approach proposed by Q12 will “benefit patients, industry, and regulatory authorities by promoting innovation and continual improvement in the pharmaceutical sector, strengthening quality control and improving supply of medicinal products” as highlighted in the guideline itself, providing that product and process knowledge are efficiently managed and combined to science- and risk- based approaches.
Leverage product and process knowledge are not only promoted by ICH Q12. Knowledge structuring is likely to become a regulatory need, as highlighted today by the FDA, with the launch of their KASA initiative3 (Knowledge-aided Assessment and Structured Applications). FDA is creating a new pharmaceutical quality assessment system, called KASA system, designed to:
1. “Capture and manage knowledge during the lifecycle of a drug product;
2.Establish rules and algorithms for risk assessment, control, and communication;
3.Perform computer-aided analyses of applications to compare regulatory standards and quality risks across applications and facilities;
4. Provide a structured assessment that minimizes text-based narratives and summarization of provided information.”
The current state and the need for the KASA system development is close to the industry state and challenges developed previously:
• Availability of previous knowledge on similar product/process: “it is not possible to easily locate historical data about similar products, processes, or the facilities. Such a practice has significantly reduced the efficiency of the regulatory assessment and increased the likelihood of inconsistencies”.
• Subjectivity in risk assessment and control, based on written “unstructured text that are not easily identifiable in lengthy assessment documents”
• “Absence of databases to capture current knowledge”…”insufficient knowledge management tools”
To face these challenges, FDA is developing KASA, a system that “captures and manages information about inherent risk and control approaches for product design, manufacturing, and facilities, in a structured format. This is intended to facilitate a concise and consistent quality assessment and largely replace freestyle text”.
Knowledge structuring and consistent quality assessment are a need for the regulatory agencies that also want to improve their assessment efficiency and consistency. Again, the need for structured databases and softwares to manage knowledge in real time (on product, process, controls and pharmaceutical activities to control quality) is raised.
Digitalization is one of the solution elements to improve the current state and answer to the identified needs. Knowledge must be available at several levels (product, process, facilities, platform knowledge, historical knowledge, current knowledge, …) and with a multivariate and multidimensional vision to facilitate converting knowledge into understanding and intelligence. This vision will be facilitated with digitalization solutions.

Conclusion
Both industry and regulators agree on the need of improved knowledge flows and risk assessments across the industry and throughout the product lifecycle. Solutions are diverse but consensus is unquestionable regarding the need for pharmaceutical industry efficiency to:
• reduce cost of development and supply,
• reduce time for product release and implementation of innovative changes,
• improve the product quality and process performance thanks to a robust control strategy and the capability to understand the relationships between process variability (CPPs) and product consistency (CQAs)
If digitalization enables a better availability and use of the knowledge, it will not improve Knowledge Management on its own. Culture, mindset evolution, people, will all be essential to use the modern KM and QRM tools efficiently.
Acknowledgments
We would like to acknowledge the following participating individuals for their contributions in the technical editing of this article:
• Yash Sabharwal, QbD Vision by CherryCircle Software
• Luke Gerrero, QbD Vision by CherryCircle Software
Partager l’article
Johanne Piriou – Aktehom
Johanne Piriou has been working for over 15 years in the pharmaceutical industry as a consultant in different operational roles notably in transfer technology projects and start-up of new facilities (C&Q, Process Validation, Process Robustness) as a specialist in process validation. She is now partner of AKTEHOM, leading the expertise in Control Strategy, QbD and Product Lifecycle Management at AKTEHOM.
johanne.piriou@aktehom.com
Michel Hertschuh – Aktehom
Michel Hertschuh has over 20 years of experience in Pharmaceutical and Medical Device industry. He is a partner and co-founder of AKTEHOM, founded in 2005. His expertise is around the aseptic operations (technical and regulatory expertise), Technology Transfer, QbD approach, Project Management, regulatory compliance and facilities design reviews. Today, Michel manages the expertise in aseptic processes and biocontamination at AKTEHOM.
Glossary
CMC Chemistry, Manufacturing & Controls
CMA Critical Material Attribute
CPP Critical Process Parameter
CQA Critical Quality Attribute
HA Health Authority
KASA Knowledge-aided Assessment and Structured Applications
KM Knowledge Management
MAH Marketing Authorization Holder
QRM Quality Risk Management
SME Subject Matter Expert
References
1. ICH Q12 – Technical and regulatory considerations for pharmaceutical product lifecycle management – Final version (Step 4) adopted on 20 November 2019
2. BioPhorum Operations Group. (March 2020):
Biomanufacturing technology roadmap – Knowledge Management
Knowledge mapping for the biopharmaceutical industry. A test case in CMC business processes from late-stage development to commercial manufacturing.
3. FDA’s new pharmaceutical quality initiative: Knowledge-aided assessment & structured applications – International Journal of Pharmaceutics (2019) – Lawrence X. Yu, Andre Raw, Larisa Wu, Christina Capacci-Daniel, Ying Zhang, Susan Rosencrance